5 Steps to Finding the Right Institutions & Principal Investigators for Phase I Trials
As pharma companies begin planning their phase I trials, they should keep patient representation goals top of mind. Finding the right institutions and principal investigators helps advance medicine for all. Here are 5 steps to achieving greater representation in clinical trials.
Step 1: Start with Inclusion
Inclusion is the precursor to building trust between patients and doctors and the bridge leading to a more inclusive healthcare system for all. Clinical operations and feasibility teams have begun to shift their approach to ensure trials are run by a more representative group of researchers.

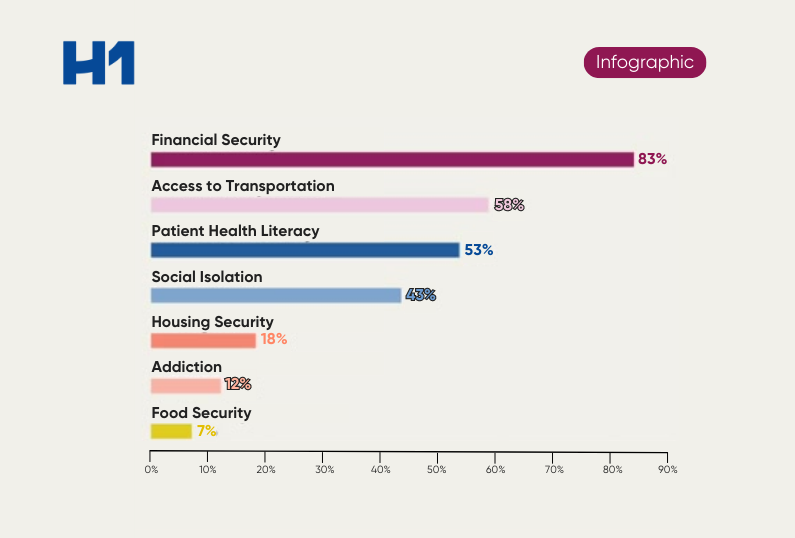
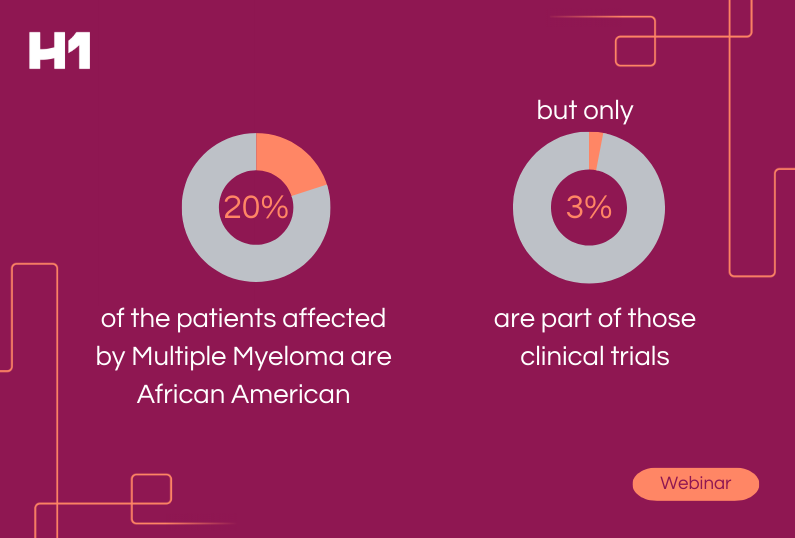
Percentage of minority groups enrolled in clinical trials as determined by JAMA Network Open study released in 2021.
Step 2: Build a Holistic Approach to Enrollment Planning
H1 uses a cross-functional approach to get a holistic understanding of trialists. By looking at patient insights generated from more than census and publicly available data, you can build more accurate profiles for Sites and Principal Investigators most likely to fit your business needs.

Step 3: Incorporate Robust Data Points

Step 4: Establish a Representation Baseline

Step 5: Visualize Data for Faster Decision Making
Layer and visualize patient, PI, and Site cohorts to support data driven decision making. Data visualization allows you to see potential PIs and sites based on location and proximity to patient clusters and much more.

Learn more about how H1 supports clinical trial representation or request a demo.