The Role of SDOH in Improving Patient Outcomes Through Clinical Trials
Social Determinants of Health (SDOH) are the conditions in which people live, learn, work, and play, which affect health access, outcomes, and overall quality of life. Collectively, they contribute significantly “to the social patterning of health, disease, and illness,” according to the Centers for Disease Control & Prevention (CDC).
A key determinant of a successful clinical study is efficiently recruiting and retaining an adequate number of the study population. However, the average enrollment efficiency is currently less than 40% for Phase 3 and 4 clinical trials. Globally, experts agree that more than 80% of trials fail to enroll on time.
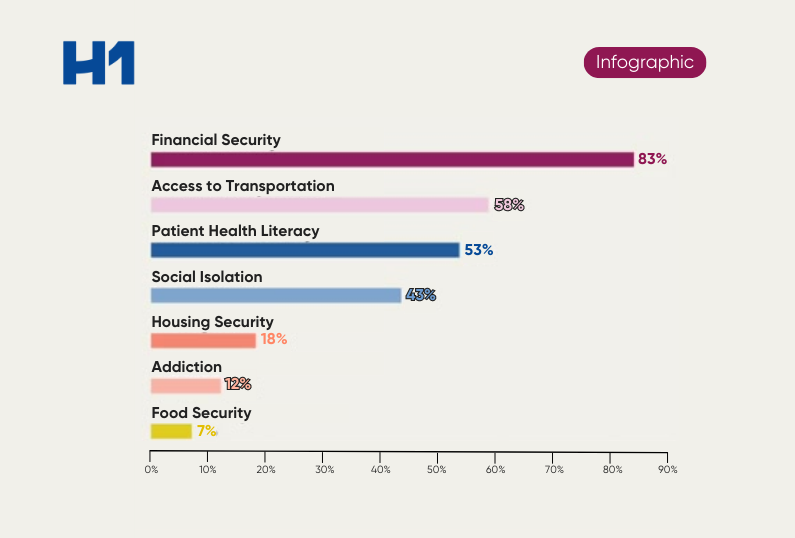
In oncology for example, financial barriers like insurance coverage or challenges with transportation have been known to impact trial participation.
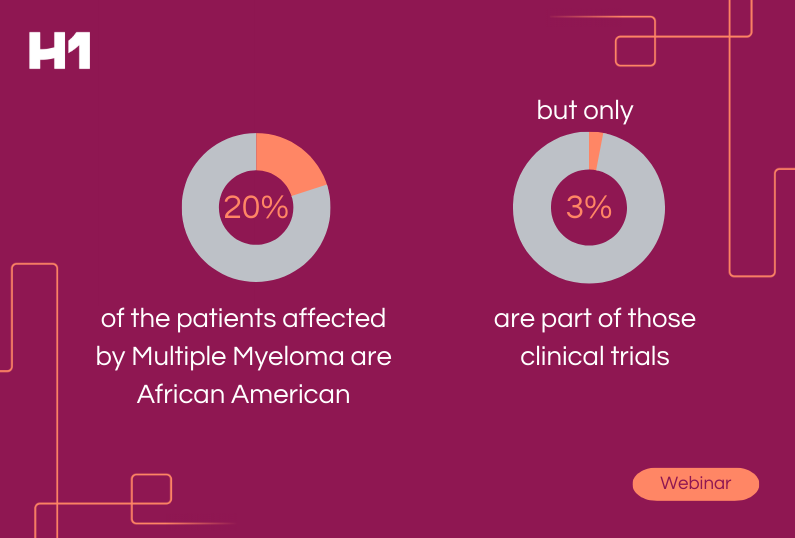
In fact, according to an article by Targeted Oncology, only 8% of cancer patients participate in clinical trials.* Biopharmaceutical companies can’t ignore that clinical trial participation is directly impacted by patient social factors and demographics. Health literacy is also a major component and one that directly affects the ability for potential participants to connect and build trust with sites and sponsors.
The Power of Trust and Inclusivity
Trust and transparency are crucial to the future of clinical trials, especially those involving underrepresented groups. Communication through trusted channels ensures that patients understand the value of their participation in clinical trials. And as it stands, the most trusted channel remains to be doctors who serve and are a part of these very communities.
Embedding health literacy into clinical trials from the beginning of the process can improve recruitment and retention. Well-established research shows that more than 72% of Americans would participate in a clinical trial if recommended by their doctor. However, only 22% said a doctor or other healthcare professional has talked to them about medical research.
Providers with established patient relationships can educate the latter about the benefits of a specific treatment or trial and explain these reasons in an understandable and approachable way.
In addition to providers, community pharmacists can be leaned on as trusted members of the healthcare industry and the community.
Recent research conducted by the Columbia University Mailman School of Public Health and Express Scripts Pharmacy found that U.S. pharmacists are highly trusted by patients and projected to play an increasingly crucial role in healthcare management. Pharmacists in ambulatory clinics, health systems, and home delivery pharmacies are at the forefront of this trend. About 80% of patients stated that they see pharmacists as an integral part of their healthcare system.
H1’s Clinical solutions features beneficial insights from potential investigators for decentralized trials to protocol development and performance analysis using data-driven decision making to help with clinical trial development, operations, feasibility, and analytics that can yield successful and insightful results for a more inclusive trial.
H1: Connecting the world with the right doctors
H1 is the connecting force for global HCP, clinical, scientific and research information. The H1 Connect platform democratizes access to HCP knowledg eand groundbreaking insights for life sciences, academic medical institutions, health systems, and payors. H1 Connect fuels a robust product that helps customers discover and engage industry experts, improve patient representation, access groundbreaking science, and accelerate commercial success with the most robust and accurate healthcare professional data.
To understand how to build more inclusive trials visit h1.co
Resources: