Data Networks & Guiding Clinical Trials
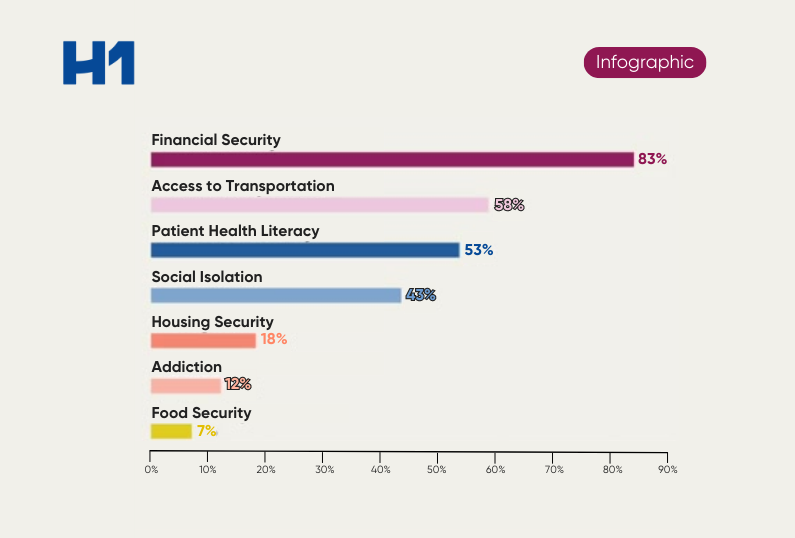
Six Metrics for Powering Trials to Reach Vulnerable Patient Populations
Join H1 industry expert, Adam Wyckoff, Senior Director of Product Management for Clinical Trials, for this informative Q&A where we pose questions about the value of data networks and powering clinical trials for success – as well as six metrics to consider when looking to meet newly updated FDA mandates and how a data partner can help.
For clinical operations and development clinical feasibility teams, patient representation leaders, as well as health economics and outcomes research (HEOR) teams, it is essential to understand the considerations when integrating data. This helps ensure more inclusive and compliant clinical trials that increase trust among patients, pharma and study participants.
Data networks are valuable to inform clinical trials with insights and analyses such as patient population selection criteria, site selection criteria, site performance evaluation criteria, Principal Investigator (PI) selection criteria, and incorporating population health efforts into informing trial selection.
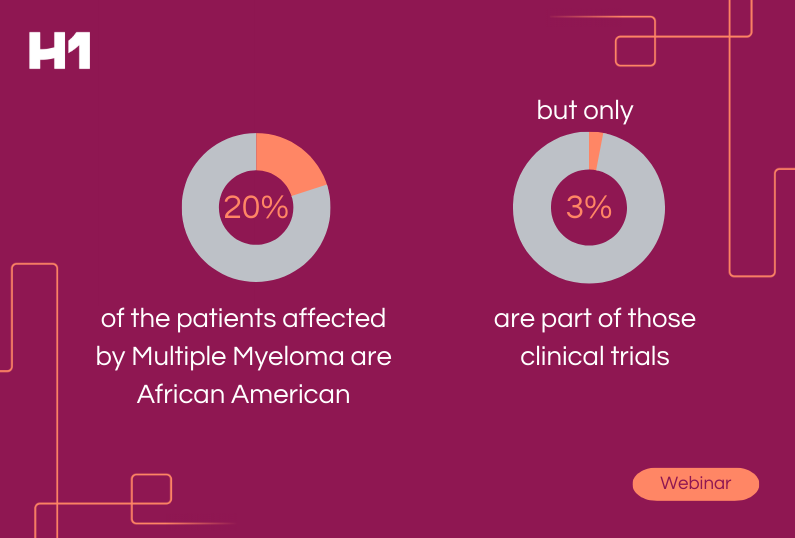
This understanding of how data networks can empower clinical trials has led to many new initiatives and regulations aimed at increasing the representation of patient representation in clinical trials.
We need to ensure that all stakeholders have access to data to inform their decision-making when it comes to inclusion in clinical trials. Therefore, data networks can be powerful for understanding disparities in access to care, which can lead to more inclusive enrollment criteria when selecting sites and PI’s for a successful trial.
Watch the video below to learn more.