The Evolution of Patient Representation in Clinical Trials
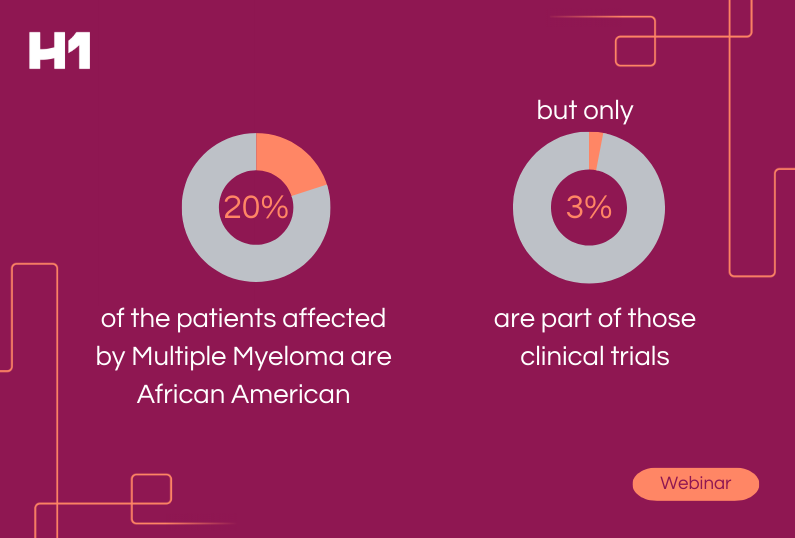
The FDA recently issued a new draft guidance for the industry about improving the enrollment of clinical trial participants from underrepresented racial and ethnic populations, many of whom are part of medically underserved communities. Improving representation in pharma will in turn improve overall patient outcomes.
“Individuals from these populations are frequently underrepresented in biomedical research despite having a disproportionate disease burden for certain diseases,” the guidance noted.
In a new two- part executive brief series, we explore:
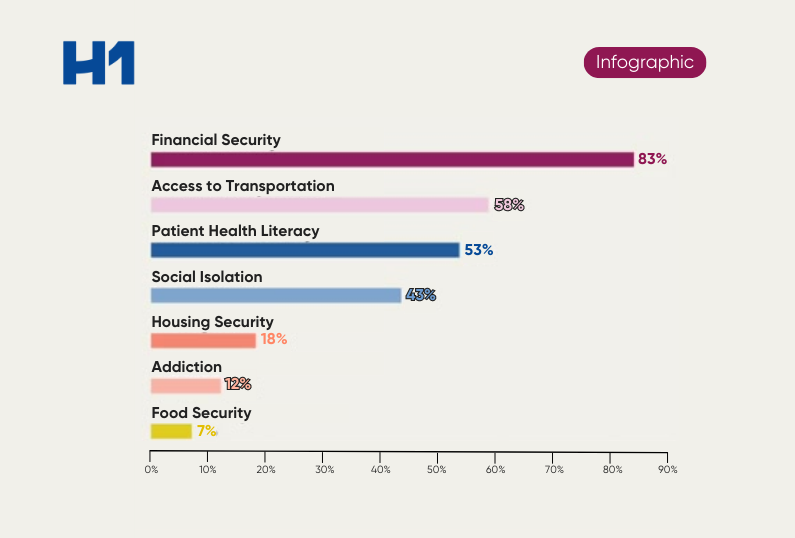
- The role of social determinants of health in building better patient representation into trials
- The critical role that trust plays in patient access and ensuring inclusive trials for drug development
H1’s Clinical solutions features beneficial insights from potential investigators for decentralized trials to protocol development and performance analysis using data-driven decision making to help with clinical trial development, operations, feasibility, and analytics that can yield successful and insightful results for a more inclusive trial.